CBSE Board Syllabus Class XI-XII
CHEMISTRY (Code No. 043)
Rationale
Higher Secondary is the most crucial stage of school education because at this juncture specialized discipline based, content-oriented courses are introduced. Students reach this stage after 10 years of general education and opt for Chemistry with a purpose of pursuing their career in basic sciences or professional courses like medicine, engineering, technology and study courses in applied areas of science and technology at tertiary level. Therefore, there is a need to provide learners with sufficient conceptual background of Chemistry, which will make them competent to meet the challenges of academic and professional courses after the higher secondary stage.
The new and updated curriculum is based on disciplinary approach with rigour and depth taking care that the syllabus is not heavy and at the same time it is comparable to the international level. The knowledge related to the subject of Chemistry has undergone tremendous changes during the past one decade. Many new areas like synthetic materials, bio-molecules, natural resources, industrial chemistry are coming in a big way and deserve to be an integral part of chemistry syllabus at senior secondary stage At international level, new formulations and nomenclature of elements and compounds, symbols and units of physical quantities floated by scientific bodies like IUPAC and CGPM are of immense importance and need to be incorporated in the updated syllabus. The revised syllabus takes care of all these aspects. Greater emphasis has been laid on use of new nomenclature, symbols and formulations, teaching of fundamental concepts, applications of concepts in chemistry to industry/ technology, logical sequencing of units, removal of obsolete content and repetition etc.
OBJECTIVES
The broad objectives of teaching Chemistry at Senior Secondary Stage are to help the learners:
to promote understanding of basic facts and concepts in chemistry while retaining the excitement of chemistry.
to make students capable of studying chemistry in academic and professional courses (such as medicine, engineering, technology) at tertiary level.
to expose the students to various emerging new areas of chemistry and apprise them with their relevance in their future studies and their application in various spheres of chemical sciences and technology.
to equip students to face various changes related to health, nutrition, environment, population, weather, industries and agriculture.
to develop problem solving skills in students.
to expose the students to different processes used in industries and their technological applications.
to apprise students with interface of chemistry with other disciplines of science such as physics, biology, geology, engineering etc.
to acquaint students with different aspects of chemistry used in daily life.
to develop an interest in students to study chemistry as a discipline.
COURSE STRUCTURE
Class XI (Theory)
-------------------------------------------------------------------------------------------------------------------------------------------------------
One Paper Time: 3 Hours 70 marks
------------------------------------------------------------------------------------------------------------------------------------------------------
Unit No. Title Marks
Unit I Some Basic concepts of chemistry 3
Unit II Structure of Atom 6
Unit III Classification of Elements and Periodicity in Properties 4
Unit IV Chemical Bonding and molecular Structure 5
Unit V States of Matter: Gases and Liquids 4
Unit VI Thermodynamics 6
Unit VII Equilibrium 6
Unit VIII Redox Reactions 3
Unit IX Hydrogen 3
Unit X S-Block Elements 5
Unit XI Some P-Block Elements 7
Unit XII Organic Chemistry: some basic Principles and Techniques 7
Unit XIII Hydrocarbons 8
Unit XIV Environmental Chemistry 3
Total : 70
-------------------------------------------------------------------------------------------------------------------------------------------------------
Unit: Some Basic Concepts of Chemistry (Periods 14)
General Introduction: Importance and scope of chemistry.
Historical approach to particulate nature of matter, laws of chemical combination. Dalton’s atomic theory: concept of elements, atoms and molecules.
Atomic and molecular masses mole concept and molar mass: percentage composition, empirical and molecular formula chemical reactions, stoichiometry and calculations based on stoichiometry.
Unit II: Structure of Atom (Periods 16)
Discovery of electron, proton and neutron; atomic number, isotopes and isobars. Thomson’s model and its limitations, Rutherford’s model and its limitations. Bohr’s model and its limitations, concept of shells and subshells, dual nature of matter and light, de Broglie’s relationship, Heisenberg uncertainty principle, concept of orbitals, quantum numbers,
shapes of s, p, and d orbitals, rules for filling electrons in orbitals - Aufbau principle, Pauli exclusion principle and Hund’s rule, electronic configuration of atoms, stability of half filled and completely filled orbitals.
Unit III: Classification of Elements and Periodicity in Properties (Periods 8 )
Significance of classification, brief history of the development of periodic table, modern periodic law and the present form of periodic table, periodic trends in properties of elements -atomic radii, ionic radii. Ionization enthalpy, electron gain enthalpy, electro negativity, valence.
Unit IV: Chemical Bonding and Molecular Structure (Periods 16)
Valence electrons, ionic bond, covalent bond: bond parameters. Lewis structure, polar character of covalent bond, covalent character of ionic bond, valence bond theory, resonance, geometry of covalent molecules, VSEPR theory, concept of hybridization, involving s, p and d orbitals and shapes of some simple molecules, molecular orbital; theory of homo nuclear diatomic molecules (qualitative idea only), hydrogen bond.
Unit V: States of Matter: Gases and Liquids (Periods 14)
Three states of matter. Intermolecular interactions, type of bonding, melting and boiling points. Role of gas laws in elucidating the concept of the molecule, Boyle’s law. Charles law, Gay Lussac’s law, Avogadro’s law. Ideal behaviour, empirical derivation of gas equation, Avogadro’s number. Ideal gas equation. Derivation from ideal behaviour,
liquefaction of gases, critical temperature.
Liquid State - Vapour pressure, viscosity and surface tension (qualitative idea only, no mathematical derivations).
Unit VI: Thermodynamics (Periods16)
Concepts Of System, types of systems, surroundings. Work, heat, energy, extensive and intensive properties, state functions.
First law of thermodynamics - internal energy and enthalpy, heat capacity and specific heat, measurement of ΔU and ΔH, Hess’s law of constant heat summation, enthalpy of: bond dissociation, combustion, formation, atomization, sublimation. Phase transformation, ionization, and solution.
Introduction of entropy as a state function, free energy change for spontaneous and nonspontaneous processes, criteria for equilibrium.
Unit VII: Equilibrium (Periods 16)
Equilibrium in physical and chemical processes, dynamic nature of equilibrium, law of mass action, equilibrium constant, factors affecting equilibrium - Le Chatelier’s principle; ionic equilibrium - ionization of acids and bases, strong and weak electrolytes, degree of ionization, concept of pH. Hydrolysis of salts (elementary idea). Buffer solutions, solubility product, common ion effect (with illustrative examples).
Unit VIII: Redox Reactions (Periods 6)
Concept of oxidation and reduction, redox reactions, oxidation number, balancing redox reactions, applications of redox reactions.
Unit IX : Hydrogen (Periods 8 )
Position of hydrogen in periodic table, occurrence, isotopes, preparation, properties and uses of hydrogen; hydrides - ionic, covalent and interstitial; physical and chemical properties of water, heavy water; hydrogen peroxide-preparation, properties and structure; hydrogen as a fuel.
Unit X: s-Block Elements (Alkali and Alkaline earth metals) (Periods 14)
Group 1 and Group 2 elements:
General introduction, electronic configuration, occurrence, anomalous properties of the first element of each group, diagonal relationship, trends in the variation of properties (such as ionization enthalpy, atomic and ionic radii), trends in chemical reactivity with oxygen, water, hydrogen and halogens; uses.
Preparation and properties of some important compounds:
Sodium carbonate, sodium chloride, sodium hydroxide and sodium hydrogen carbonate, biological importance of sodium and potassium.
CaO, CaCO3 and industrial use of lime and limestone, biological importance of Mg and Ca
Unit XI: Some p-Block Elements (Periods 16)
General Introduction to p-Block Elements
Group 13 elements: General introduction, electronic configuration, occurrence. Variation of properties, oxidation states, trends in chemical reactivity, anomalous properties of first element of the group; Boron- physical and chemical properties, some important compounds:
borax, boric acids, boron hydrides. Aluminium: uses, reactions with acids and alkalies.
Group 14 elements: General introduction, electronic configuration, occurrence, variation of properties, oxidation states, trends in chemical reactivity, anomalous behaviour of first element, Carbon - catenation, allotropic forms, physical and chemical properties; uses of some important compounds: oxides.
Important compounds of silicon and a few uses: silicon tetrachloride, silicones, silicates and zeolites.
Unit XII: Organic Chemistry - Some Basic Principles and Techniques (Periods 14)
General introduction, methods of qualitative and quantitative analysis, classification and IUPAC nomenclature of organic compounds
Electronic displacements in a covalent bond: inductive effect, electromeric effect, resonance and hyper conjugation.
Homolytic and heterolytic fission of a covalent bond: free radicals, carbocations, carbanions; electrophiles and nucleophiles, types of organic reactions
Unit XIII: Hydrocarbons (Periods 16)
Classification of hydrocarbons
Alkanes - Nomenclature, isomerism, conformations (ethane only), physical properties, chemical reactions including free radical mechanism of halogenation, combustion and pyrolysis.
Alkenes - Nomenclature, structure of double bond (ethene) geometrical isomerism, physical properties, methods of preparation; chemical reactions: addition of hydrogen, halogen, water, hydrogen halides (Markovnikov’s addition and peroxide effect), ozonolysis, oxidation, mechanism of electrophilic addition.
Alkynes - Nomenclature, structure of triple bond (ethyne), physical properties. Methods of preparation, chemical reactions: acidic character of alkynes, addition reaction of - hydrogen, halogens, hydrogen halides and water.
Aromatic hydrocarbons: Introduction, IUPAC nomenclature; benzene: resonance aromaticity; chemical properties: mechanism of electrophilic substitution. – nitration sulphonation, halogenation, Friedel Craft’s alkylation and acylation: directive influence of functional group in mono-substituted benzene; carcinogenicity and toxicity.
Unit XIV: Environmental Chemistry (Periods 6)
Environmental pollution - air, water and soil pollution, chemical reactions in atmosphere, smog, major atmospheric pollutants; acid rain, ozone and its reactions, effects of depletion of ozone layer, greenhouse effect and global warming - pollution due to industrial wastes; green chemistry as an alternative tool for reducing pollution, strategy for control of
environmental pollution.
Practicals
-----------------------------------------------------------------------------------------------------------------------------------------------
Evaluation Scheme for Examination Marks
Volumetric Analysis 10
Salt Analysis 6
Content Based Experiment 4
Class Record and Viva 5
Investigatory project 5
Total 30
-----------------------------------------------------------------------------------------------------------------------------------------------
PRACTICALS SYLLABUS Total Periods 60
A. Basic Laboratory Techniques (Periods 2)
1. Cutting glass tube and glass rod
2. Bending a glass tube
3. Drawing out a glass jet
4. Boring a cork
B. Characterization and purification of chemical substances (Periods 6)
1. Determination of melting point of an organic compound
2. Determination of boiling point of an organic compound
3. Crystallization of impure sample of anyone of the
following: Alum, copper sulphate, Benzoic acid.
C. Experiments related to pH change (Periods 6)
(a) Anyone of the following experiments:
Determination of pH of some solutions obtained from fruit juices, varied concentrations of acids. ,bases and salts using pH paper or universal indicator.
Comparing the pH of solutions of strong and weak acid of same concentration.
Study the pH change in the titration of a strong base using universal indicator.
b) Study of pH change by common-ion effect in case of weak acids and weak bases.
D. Chemical equilibrium (Periods 4)
One of the following experiments:
(a) Study the shift in equilibrium between ferric ions and thiocyanate ions by increasing/ decreasing the concentration of either ions.
(b) Study the shift in equilibrium between [Co(H2O)6]2+ and chloride ions by changing the concentration of either of the ions.
E. Quantitative estimation (Periods 16)
Using a chemical balance.
Preparation of standard solution of oxalic acid.
Determination of strength of a given solution of sodium hydroxide by titrating it against standard solution of oxalic acid.
Preparation of standard solution of sodium carbonate.
Determination of strength of a given solution of hydrochloric acid by titrating it against standard sodium carbonate solution.
F. Qualitative analysis (Periods 16)
Determination of one anion and one cation in a given salt
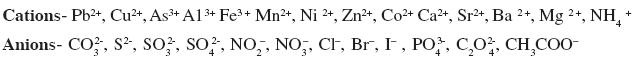
(Note: Insoluble salts excluded)
G. Detection of nitrogen, sulphur, Chlorine (Periods 10)
bromine and iodine in an organic compound.
PROJECT (Periods 10)
Scientific investigations involving laboratory testing and collecting information from other sources.
A Few suggested Projects
Checking the bacterial contamination in drinking water by testing sulphide ion.
Study of the methods of purification of water.
Testing the hardness, presence of iron, fluoride, chloride etc. depending upon the regional variation in drinking water and the study of causes of presences of these ions above permissible limit (if any).
Investigation of the foaming capacity of different washing soaps and the effect of addition of sodium carbonate on them.
Study of the acidity of different samples of the tea leaves.
Determination of the rate of evaporation of different liquids.
Study of the effect of acids and bases on the tensile strength of fibers.
Analysis of fruit and vegetable juices for their acidity.
Note: Any other investigatory project, which involves about 10 period of work, can be chosen with the approval of the teacher.
Recommended Textbooks.
1. Chemistry Part - I, Published by NCERT
2. Chemistry Part - II, Published by NCERT
COURSE STRUCTURE
Class XII (Theory)
-------------------------------------------------------------------------------------------------------------------------------------------------------
One Paper Time: 3 Hours 70 marks
------------------------------------------------------------------------------------------------------------------------------------------------------
Unit No. Title Marks
Unit II Solutions 5
Unit III Electrochemistry 5
Unit IV Chemical kinetics 5
Unit V Surface chemistry 4
Unit VI General principles and processes of Isolation of Elements 3
Unit VII p-Block Elements 8
Unit VIII d- and f- Block Elements 5
Unit IX Coordination Compounds 3
Unit X Haloalkanes and Haloarenes 4
Unit XI Alcohols, Phenols and Ethers 4
Unit XII Aldehydes, Ketones and Carboxylic acids 6
Unit XIII Organic Compounds containing Nitrogen 4
Unit XIV Biomolecules 4
Unit XV Polymers 3
Unit XVI Chemistry in Everyday life 3
Total : 70
-------------------------------------------------------------------------------------------------------------------------------------------------------
Unit I: Solid State (Periods 12)
Classification of solids based on different binding forces: molecular, ionic, covalent and metallic solids, amorphous and crystalline solids (elementary idea), unit cell in two dimensional and three dimensional lattices, calculation of density of unit cell, packing in solids, voids, number of atoms per unit cell in a cubic unit cell, point defects, electrical and magnetic properties.
Unit II: Solutions (Periods 12)
Types of solutions, expression of concentration of solutions of solids in liquids, solubility of gases in liquids, solid solutions, colligative properties – relative lowering of vapour pressure, elevation of Boiling Point, depression of freezing point, osmotic pressure, determination of molecular masses using colligative properties, abnormal molecular mass.
Unit III: Electrochemistry (Periods 14)
Redox reactions, conductance in electrolytic solutions, specific and molar conductivity variations of conductivity with concentration, Kohlrausch’s Law, electrolysis and laws of electrolysis (elementary idea), dry cell – electrolytic cells and Galvanic cells; lead accumulator, EMF of a cell, standard electrode potential, Nernst equation and its application to chemical cells, fuel cells; corrosion.
Unit IV: Chemical Kinetics (Periods 12)
Rate of a reaction (average and instantaneous), factors affecting rate of reaction; concentration, temperature, catalyst; order and molecularity of a reaction; rate law and specific rate constant, integrated rate equations and half life (only for zero and first order reactions); concept of collision theory (elementary idea, no mathematical treatment)
Unit V: Surface Chemistry (Periods 8 )
Adsorption – physisorption and chemisorption; factors affecting adsorption of gases on solids; catalysis : homogenous and heterogeneous, activity and selectivity: enzyme catalysis; colloidal state: distinction between true solutions, colloids and suspensions; lyophilic, lyophobic, multimolecular and macromolecular colloids; properties of colloids; Tyndall effect, Brownian movement, electrophoresis, coagulation; emulsion – types of emulsions.
Unit VI: General Principles and Processes of Isolation of Elements (Periods 8 )
Principles and methods of extraction - concentration, oxidation, reduction electrolytic method and refining; occurrence and principles of extraction of aluminium, copper, zinc and iron.
Unit VII: p-Block Elements (Periods 14)
Group 15 elements: General introduction, electronic configuration, occurrence, oxidation states, trends in physical and chemical properties; nitrogen - preparation, properties and uses; compounds of nitrogen: preparation and properties of ammonia and nitric acid, oxides of nitrogen (structure only); Phosphorous-allotropic forms; compounds .of phosphorous: preparation and properties of phosphine, halides (PCl3, PCl5) and oxoacids (elementary idea only)
Group 16 elements: General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; dioxygen: preparation, properties and uses; simple oxides; Ozone. Sulphur - allotropic forms; compounds of sulphur: preparation, properties and uses of sulphur dioxide; sulphuric acid: industrial process of manufacture, properties and uses, oxoacids of sulphur (structures only).
Group 17 elements: General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; compounds of halogens: preparation, properties and uses of chlorine and hydrochloric acid, interhalogen compounds, oxoacids of halogens (structures only).
Group 18 elements: General introduction, electronic configuration. Occurrence, trends in physical and chemical properties, uses.
Unit VIII: d and f Block Elements (Period 14)
General introduction ,electronic configuration, occurrence and characteristics of transition metals, general trends in properties of the first row transition metals – metallic character, ionization enthalpy, oxidation states, ionic radii, colour catalytic property, magnetic properties, interstitial compounds, alloy formation preparation and properties of K2Cr2O7 and KMnO4.
Lanthanoids - electronic configuration, oxidation states, chemical reactivity and lanthanoid contraction.
Actinoids - Electronic configuration, oxidation states.
Unit IX: Coordination Compounds (Period 12)
Coordination compounds - Introduction, ligands, coordination number, colour, magnetic properties and shapes, IUPAC nomenclature of mononuclear coordination compounds. bonding; isomerism, importance of coordination compounds (in qualitative analysis, extraction of metals and biological systems).
Unit X: Haloalkanes and Haloarenes. (Periods 12)
Haloalkanes:
Nomenclature, nature of C-X bond, physical and chemical properties, mechanism of substitution reactions.
Haloarenes:
Nature of C-X bond, substitution reactions (directive influence of halogen for monosubstituted compounds only)
Uses and environmental effects of - dichloromethane, trichloromethane, tetrachloromethane, iodoform, freons, DDT.
Unit XI: Alcohols, Phenols and Ethers (Periods 12)
Alcohols: Nomenclature, methods of preparation, physical and chemical properties (of primary alcohols only); identification of primary, secondary and tertiary alcohols; mechanism of dehydration, uses of methanol and ethanol.
Phenols : Nomenclature, methods of preparation, physical and chemical properties, acidic nature of phenol, electrophillic substitution reactions, uses of phenols.
Ethers: Nomenclature, methods of preparation, physical and chemical properties, uses.
Unit XII: Aldehydes, Ketones and Carboxylic Acids (Periods 12)
Aldehydes and Ketones: Nomenclature, nature of carbonyl group, methods of preparation, physical and chemical properties mechanism of nucleophilic addition, reactivity of alpha hydrogen in aldehydes; uses.
Carboxylic Acids: Nomenclature, acidic nature, methods of preparation, physical and chemical properties; uses.
Unit XIII: Organic compounds containing Nitrogen (Periods 10)
Amines: Nomenclature, classification, structure, methods of preparation, physical and chemical properties, uses, identification of primary, secondary and tertiary amines.
Cyanides and Isocyanides - will be mentioned at relevant places in context.
Diazonium salts: Preparation, chemical reactions and importance in synthetic organic chemistry.
Unit XIV: Biomolecules (Periods 12)
Carbohydrates - Classification (aldoses and ketoses), monosaccahrides (glucose and fructose), oligosaccharides (sucrose, lactose, maltose), polysaccharides (starch, cellulose, glycogen); importance.
Proteins - Elementary idea of α - amino acids, peptide bond, polypeptides, proteins, structure of amines-primary, secondary, tertiary structure and quaternary structures (qualitative idea only), denaturation of proteins; enzymes.
Vitamins -Classification and functions.
Nucleic Acids: DNA and RNA .
Unit XV: Polymers (Periods 8 )
Classification - natural and synthetic, methods of polymerization (addition and condensation), copolymerization. Some important polymers: natural and synthetic like polythene, nylon, polyesters, bakelite, rubber.
Unit XVI: Chemistry in Everyday life: (Period 8 )
1. Chemicals in medicines - analgesics, tranquilizers, antiseptics, disinfectants, antimicrobials, antifertility drugs, antibiotics, antacids, antihistamines.
2. Chemicals in food - preservatives, artificial sweetening agents.
3. Cleansing agents - soaps and detergents, cleansing action.
Practicals
-----------------------------------------------------------------------------------------------------------------------------------------------
Evaluation Scheme for Examination Marks
Volumetric Analysis 10
Salt Analysis 6
Content Based Experiment 4
Class Record and Viva 5
Investigatory project 5
Total 30
-----------------------------------------------------------------------------------------------------------------------------------------------
PRACTICALS SYLLABUS Total Periods 60
A. Surface Chemistry. (Periods 6)
(a) Preparation of one lyophilic and one lyophobic sol.
Lyophilic sol - starch, egg albumin and gum
Lyophobic sol - aluminium hydroxide, ferric hydroxide, arsenous sulphide.
(b) Study of the role of emulsifying agents in stabilizing the emulsions of different oils.
B. Chemical Kinetics (Periods 4)
(a) Effect of concentration and temperature on the rate of reaction between sodium thiosulphate and hydrochloric acid.
(b) Study of reaction rates of any one of the following:
(i) Reaction of iodide ion with hydrogen peroxide at room temperature using different concentration of iodide ions.
(ii) Reaction between potassium iodate, KIO3 and sodium sulphite: (Na2SO3) using starch solution as indicator (clock reaction).
C. Thermochemistry (Periods 4)
Any one of the following experiments
i) Enthalpy of dissolution of copper sulphate or potassium nitrate.
ii) Enthalpy of neutralization of strong acid (HC1) and strong base (NaOH)
iii) Determination of enthalpy change during interaction (Hydrogen bond formation) between acetone and chloroform
D. Electrochemistry (Period 2)
Variation of cell potential in Zn/Zn2+||Cu2+/Cu with change in concentration of electrolytes (CuSO4 or ZnSO4) at room temperature.
E. Chromatography (Periods 2)
i) Separation of pigments from extracts of leaves and flowers by paper chromatography and determination of Rf values.
ii) Separation of constituents present in an inorganic mixture containing two cations only (constituents having large difference in Rf values to be provided).
F. Preparation of Inorganic Compounds (Periods 4)
i) Preparation of double salt of ferrous ammonium sulphate or potash alum.
ii) Preparation of potassium ferric oxalate.
G. Preparation of Organic Compounds (Periods 4)
Preparation of any two of the following compounds
i) Acetanilide
ii) Di-benzal acetone
iii) p-Nitroacetanilide.
iv) Aniline yellow or 2 - Napthol aniline dye.
v) Iodoform
H. Tests for the functional groups present in organic compounds: (Periods 6)
Unsaturation, alcoholic, phenolic, aldehydic, ketonic, carboxylic and amino (primary) groups.
I. Characteristic tests of carbohydrates, fats and proteins in pure samples and their detection in given food stuffs. (Periods 4)
J. Determination of concentration/molarity of KMnO4 solution by titrating it against a standard solution of: (Periods 8 )
i) Oxalic acid,
ii) Ferrous ammonium sulphate (Students will be required to prepare standard solutions by weighing themselves).
K. Qualitative analysis (Periods 14 )
Determination of one cation and one anion in a given salt.
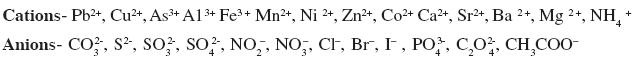
(Note: Insoluble salts excluded)
PROJECT
Scientific investigations involving laboratory testing and collecting information from other sources.
A few suggested Projects.
Study of presence of oxalate ions in guava fruit at different stages of ripening.
Study of quantity of casein present in different samples of milk.
Preparation of soybean milk and its comparison with the natural milk with respect to curd formation, effect of temperature, etc.
Study of the effect of potassium bisulphate as food preservative under various conditions (temperature, concentration, time etc.) :
Study of digestion of starch by salivary amylase and, effect of pH and temperature on it.
Comparative study of the rate of fermentation of following materials: wheat flour, gram flour, potato juice, carrot juice etc.
Extraction of essential oils present in Saunf (aniseed), Ajwain (carum), Illaichi (cardamom).
Study of common food adulterants in fat, oil, butter, sugar, turmeric powder, chilli powder and pepper.
Note: Any investigatory project, which involves about 10 periods of work, can be chosen with the approval of the teacher.
Recommended Textbooks.
1. Chemistry Part - I, Published by NCERT
2. Chemistry Part - II, Published by NCERT